Protecting the Ozone Layer
Every spring in the Southern Hemisphere since the late 1970's, scientists have observed a depletion of the ozone layer over Antarctica. The region of decreased ozone is known as the ozone hole. Eventually, scientists figured out the chemistry of stratosphereic ozone and realized it was at risk as a result of chlorofluorocarbons (CFCs) used as propellants and refrigerants. A smaller ozone hole developes annually over the northern hemisphere, and the average ozone concentrations have been reduced somewhat at all latitudes.

People make and emit chemicals such as chlorofluorocarbons (CFCs). CFCs are very stable in the troposphere. They move to the stratosphere and are broken down by strong ultraviolet (UV) radiation, where they release chlorine atoms that then deplete the ozone layer. Without the ozone to absorb UV radiation (or UVB) in the stratosphere, much more UVB can reach the ground, leading to skin cancer and disruption of plant and animal species. This is a main source of concern about the ozone hole and CFCs.
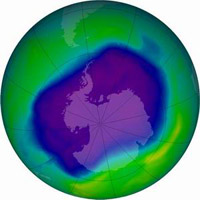
In the late 20th century, leaders for many of the world's countries got together in Montreal, Canada, and agreed to stop producing and emitting CFCs. This 'Montreal Protocol' is one of the greatest success stories of all time, because people became aware of a serious environmental problem, addressed it politically, and found a solution. While the hole in the ozone has not yet recovered to it's preindustrial levels, stratospheric ozone is not decaying nearly as fast as it had been in the 20th century. In the image above, the blue area has one-third less ozone than the green area.
DID YOU KNOW?
A similar protocol for greenhouse gases was subsequently drawn in Kyoto, but it has not met with much success. This may be due to a current lack of cheap and easy replacements for fossil fuels to produce energy for modern society.
